
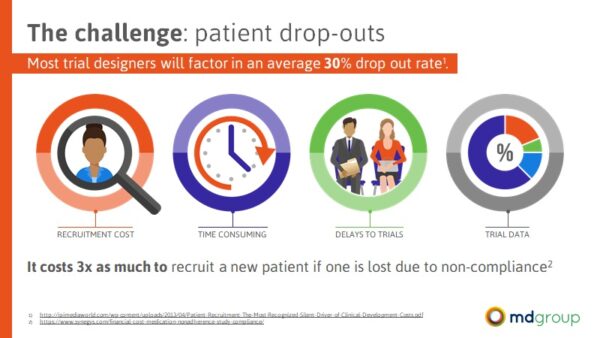
Patient retention is a huge issue when it comes to clinical trial success, and some trial designers will even factor in a dropout rate as high as 30% when budgeting and planning for a trial.
In the third of our series of mdWebinars on July 9, our panel discussed the reasons why patients drop out and what can be done to reduce the burden of trial participation for patients, and improve patient engagement and retention.
Our moderator facilitated discussion and debate among our panellists:

Andrew Schorr, Co-founder and President at Patient Power LLC
Andrew Schorr is co-founder of PatientPower.info and living with two blood cancers. He has been in two clinical trials, one that saved his life, and an another that discovered a second cancer early and matched him with a newly approved medicine. He is very grateful. Andrew’s team reaches more than 250,000 people affected by cancer each month. Many express interest in trials and become strong advocates for the full range of treatment opportunities that could be right for them.
Carole Scrafton, Co-founder and Chair, FibroFlutters
Carole Scrafton is CEO of fibroflutters.com and Global Social Media Network & Co-founder and Founding Five member of FibroFlutters Community Support. Carole is a Health, Patient, Research & Pharma advocate related to chronic /invisible/rare illnesses; Patient Partner with PFMD & Freelance researcher with Envision Pharma Group; and a patient speaker & author. She advocates for a multi-disciplinary approach to all aspects of medical healthcare and for better clinical trial education & processes for people with chronic/invisible/rare illnesses and multiple chronic conditions, where an all-stakeholder approach is utilised to create better outcomes for all, not just patients.
Nuala Ryan, Clinical Research Consultant
Nuala Ryan has experience as both a cancer patient and in the clinical trial industry, so has a perspective from both sides. She is a graduate of the Irish Platform for Patient Organisation Science and Industry (IPPOSI) Patient Education course, part of the Irish National EUPATI Platform, participated in research trials and has numerous patient advisory boards and panels. She also has 27 years of experience working in clinical research in Contract Research Organisations.
We opened the discussion with the implications of patient drop-outs from clinical trials including recruitment costs, time, delays to trials, and data quality, highlighting that “it costs 3 times as much to recruit a new patient to replace one that dropped out.”
We then posed the first question to the panel:
From your personal and/or professional experience, what are some of the challenges that patients face when participating in trials?
Andrew Schorr highlighted the need to “treat patients as investors, not guinea pigs” in clinical trials. He talked about the need for patients to get updates on the progress of the trial generally and not just about themselves; and to be shown appreciation for the difference a patient’s participation makes/has made – “patients are pioneers” helping with research to bring new treatments to others.
Carole Scrafton talked about the issue of awareness and education. People don’t know about the clinical trial process or even where to find clinical trials. They don’t understand the language and jargon. And if they don’t understand trials then they won’t apply to participate in them. One of the biggest challenges Carole sees and has experienced herself is the travel to the hospital/clinic. Patients can be physically and mentally exhausted by travelling to/from their appointments.
We then asked the audience:
What are you seeing as the biggest reason for patients dropping out of a trial?
- Travel difficulties
- Financial constraints
- Fear/anxiety
- Schedule conflicts/forgetting visits
- Lack of appreciation
47% said ‘Travel difficulties’, with ‘Lack of appreciation chosen by 20%, followed by ‘Fear/anxiety’ and ‘Schedule conflicts/forgetting visits’ both at 13%.
There was also an excellent point from the audience that there was no option related to the challenge of communications. As identified by the panel, a lack of or poor or confusing communications is also a reason why patients may drop out of a trial.
The second question was posed:
What initiatives have you seen the industry taking to ease the burden of trial participation?
Nuala Ryan highlighted industry initiatives to increase patient involvement upfront, during and after the trial. Upfront, to include patients and sites in protocol reviews workshops to ensure trials are designed from the outset with patients in mind. During the study to ensure patients are well supported with travel and logistics and kept engaged through regular, clear communications. And that after the trial has closed, there is follow-up to show appreciation for the patient’s contribution to the research, and updates on the study results.
Andrew and Carole followed up with their views on industry initiatives before Vale moved onto the final question of what the panel would like to see the industry do in the future to improve the patient experience.
To hear what they had to say, you can watch the full replay below:









