
There’s no doubt of the seismic impact COVID-19 has had on clinical trials. As a result of safety measures, the industry has had no choice but to start moving towards an approach which minimises or eliminates the need for travel and contact. As a result, more and more trials are being held virtually, and completely decentralised trials are increasingly more common.
While this is a step in the right direction towards truly patient-centric trials, there’s still work to be done. We’ve already seen how reducing patient burden in decentralised trials can boost recruitment and retention rates, but involving patients in the design of these trials can be beneficial in many ways too. Especially when CROs and healthcare providers are still navigating their way through new virtual and digital methods.
In this article, we’ll look further into the necessity of involving patients in the end to end design of trials, the catalytic effects of COVID-19 on decentralisation, and how we can implement practices to steer the clinical research process to become more patient-focused.
What COVID-19 has taught us about patient-focused trials
While elements of digital innovation have been explored in clinical trials for some time, virtual trials had been somewhat of a “novel idea” – until the COVID-19 pandemic.
In our mdWebinar earlier this year on the lessons learned from the pandemic, Rhonda Henry, Vice President at Patient Centered Trials – PPD, said: “Pre-COVID, we found that the willingness of customers or sponsors to entertain things like TeleVisits, remote eConsent and home health care – much less a fully digital trial – were interesting conversations, but they seemed too innovative to be adopted in the majority of trials.
“Fast forward to COVID, and we quickly learned there’s a cost behind not being innovative enough. Out of necessity, we’ve had to adopt many of these solutions if for no other reason than to stay connected to our patients, to keep that physician and patient relationship in a time when everyone is isolated.”
Adapting to fully virtual trials and realising their full impact on patient happiness will require a continued commitment from study organisers. As Henry highlighted, it’s important that the industry doesn’t forgo the progress made towards patient-centricity, and slip back into its old ways once travel and contact restrictions are lifted.
She added: “We have to look at how we’re designing our clinical trials. It has to be the right patient population, with the right endpoints, incorporating the right technology.
“We need to recognise that even though travel restrictions may be lifted, there are going to be patients and caregivers that are reluctant to get back out into the community to go to a medical centre. The onus is on sites to really understand their patients and what their concerns are, and mitigate the risks of losing patients because they don’t want to leave their home, or because there are other socioeconomic factors involved.”
Why patients need to be involved in clinical trial design
We know the most common reasons patients cite for dropping out of clinical trials. How many of these issues might have been avoided if patients had more input in informing the end to end design?
According to the UK’s National Institute of Health Research, involving patients from the beginning of the clinical development process has the potential to significantly improve study design and delivery, resulting in better recruitment, retention, and protocol adherence.
In our first mdWebinar Nick Hicks, Director of Commutateur Advocacy Communications, highlighted that companies who already adopt a strong patient-centric culture will have a competitive advantage as the industry transitions to a “new normal”.
This was echoed across our entire webinar series this year, with almost all panellists agreeing that patient voices need to be present during design discussions so the industry can better understand their needs and expectations of the research process.
To do this, sponsors and CROs will need to work closely with patient advocates, patient groups and informed patients. By bringing these groups in as consultants and partners, study organisers can better understand and address their participants’ concerns and are equipped to support them through the entire clinical trial process.
Involving different patients at different stages of clinical trial development
Patients shouldn’t only be involved in trial logistics such as travel, site visits or technology use – but from the very start of the research process. Gaining patient insight at every step of the way not only helps them feel valued, heard, and supported but offers an essential perspective on your trial design. After all, who better knows about a patient’s needs and experiences than patients themselves?
However, the right patient groups must be involved at the right stages, as patient advocate Trishna Bharadia explained in our mdWebinar on patient recruitment and retention: “When patients are becoming involved in the process itself, outside of purely being a participant, the industry needs to make sure they’re getting the right types of patients involved at the right times.”
There will be times when it’s beneficial to speak to ‘expert’ patients and highly informed advocacy groups, and times when it’s vital to speak to patients who don’t have as much knowledge of their condition, general health literacy, or advocacy experience.
Bharadia explained that there are broadly three groups of patients:
- Patients by experience who know their own experience with their condition, and can offer feedback on elements of the trial process such as travel and logistics which have a direct impact on their lives
- Expert patients who also know a lot about the experiences of the patient community and issues affecting them, and can anticipate other patients’ needs and share anecdotal advice from theirs and others’ experiences
- Pro patients who know different patient communities, are aware of issues affecting the healthcare system and can offer advice on the entire journey from recruitment through to completion
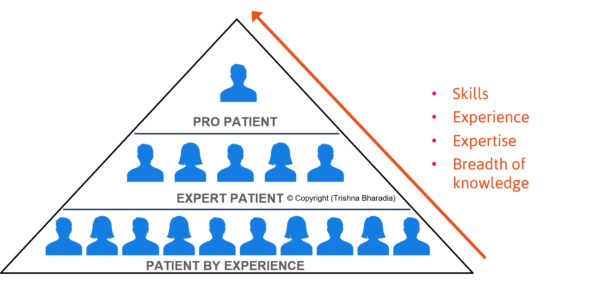
Patient Experience Pyramid – © Copyright Trishna Bharadia
Involving patients in clinical trial design effectively
Consulting with patients on the clinical trial journey is of course only the first step, then study designers must ensure patient input is integrated into trial design.
As Laree Tracy explained to us in her mdWebinar: “The challenge is getting whatever [patients] communicate back to the study designers to ensure that the endpoint is translated into what the patient’s asking for. There’s a gap there – it’s a lot about health literacy on the patient’s behalf.”
To bridge the disconnect between gathering patient input and making changes to study design, sponsors and CROs must work to cultivate trust with patients, see the bigger picture, and be willing to increase their budgets for a longer-term gain. They may need to create more formal patient-led roles within their teams, and ensure the consultancy process allows for additional rounds of feedback to translate patient input into the trial endpoints.
In our mdWebinar on integrating technology in clinical trials Victoria Siegrist, Global Patient Partnership Director at Roche, said: “We shouldn’t stop at the stage of listening to what participants actually need. We should also co-create with them and collaborate with them.”
The future of clinical trials calls for a cooperative approach with patients
The effects of COVID-19 on the research industry has posed huge challenges in clinical trial development to execution. However, the shift to more decentralised and virtual trials has opened up a huge opportunity and highlighted the necessity for patient engagement in the entire trial design process.
Clinical trials cannot claim to consider the patients’ best interests without consulting them throughout the different stages of the design process. Nobody knows about their experience better than they do.
When patients’ views, advice and experiences are successfully incorporated into trial design, patients feel more in control of their participant and supported on a deeper level, allowing clinical trials to function more efficiently, with increased patient retention, delivering faster and better results.
Think remarkable. Expect remarkable. BE remarkable.
We deliver remarkable patient experiences that improve clinical trial outcomes.
We empower healthcare providers with next-generation care management tools and technologies.
We help CROs deliver high-performance patient experience management, and support pharma / biotech to attract, engage, and maintain patient participation in clinical trials.
Remarkable means the right people, the right technologies and the right systems to deliver the best possible outcomes for you, your patients, and your clinical trial.
Let’s make your next study remarkable. Contact us now to discuss how.









